(c) 2006 Brian Altonen
Non-HEDIS Prescription Drug Measures
The design of a Non-HEDIS related measure of prescription drugs utilization focused on three approaches to the data:
- the chronic use of drugs by particular disease/patient types due to ongoing high costs associated with these illnesses; these illnesses are typically not reviewed as a part of HEDIS.
- the acute to moderate use of high cost medications for which cost-benefits reviews have to be considered and the overall effectiveness of the medication reviewed in the literature and taken into account
- the use of drugs related to iatrogenic causes or illnesses that are somehow otherwise considered highly preventable should an effective means of intervention be developed.
As noted elsewheere in this report, this activity was in part engaged in with the goal of determining if exceptionally high costs existed for some individuals (such cases were in fact founded and due to institutional and program-related causes were appropriately dealt with), and whether or not indicators of addiction and overuse could be found. (This latter administrative concern was no longer the case following my review.)
Examples of the first class of scripts to be focused upon include drugs administered to patients with RA, MS, GERD, Fibromyalgia, Migraine, Chronic Insomnia, Long-term Infectious Diseases, Parkinsonism, Alzheimer’s, etc. The second class of scripts involve high cost cancer remedies, interferon, Monoclonal Antibodies, high cost anti-bacterials, anti-virals, etc. The third class of drugs are those used primarily within the hospital environment in order to treat iatrogenic and Autoimmune deficiency related conditions, or those used to treat individuals with a history of what is considered a highly preventable condition such as certain STDs, pre-diabetic to NIDDM cases, Emergent/Urgent High Risk asthmatic cases with a history of non-steroidal or non-anti-inflammatory drug use, Chronic Heart Disease with inadequate multisystems monitoring engaged in by the PCP, etc.
The best way to review these conditions is to base the order of review on age group associations. From the review of HEDIS activities that can be related to script behaviors, we know that the 18 to 65 yo age group has a certain degree of adequate coverage, although additional reviews need to be added in order to make sure that all disease groups are reviewed for such a population health measure. The <18 yo group has STDs, childhood disease onset (leukemia, type 1 DM, hyperallergenesis, early onset autoimmune diseases, genetic diseases) and several infectious disease classes in need of monitoring. For the remaining age range in the research population if a systems approach is used to identify particular disease types that could be used as indicators of change, then a method of monitoring population health can be developed that makes use of prescription data in combination with the ICD data that is available.
By developing a better understanding of correlations expected between a given ICD and the drug itself, we can use that understanding of this particular relationship to define the next step in this research. In my study of chemical spills in relation to Cancer for example, a GIS project carried out from ca. 1999 to 2008 approximately (but still ongoing at times), I was able to develop profiling methods for documenting counts and types/groups of chemicals used in relation to particular SIC (Standard Industrial Classification) classes. Applying a similar methodology, by substituting drug class information for chemical groups, and ICD- or pharma-related disease classes for SIC, a method of comparing these two groups could be developed using some form of matrix-based, covariance or correlation based evaluation of the combinations of findings for each group of drugs, per disease type (based on class), and vice versa. The next step would be to determine which of these two methods works better in the real pharma environment.
One of the disadvantages to reviewing script data is that protocols are constantly updating and changing. This is one such reason why such a use of the script-claims data may help in defining profiles for a particular set of illnesses, for a particular age group, under the care of a given health care provider or insurance agency. The updates will have less of an impact of the methodology itself assuming its formulas or algorithms are produced in a broad-enough fashion. This use of a broad-classification technique also eliminated the statistical problems that can ensue due to small group analyses. By combining certain disease types together as was done in the HEDIS measure “Annual Monitoring for Patients on Persistent Medications”, one can produce a statistically viable measurement tool that can depict when statistically significant outcomes are obtained. Since this is a multitopic study with several diseases merged together to form the research group (i.e. you decide to review epilepsy and MS together as a group to increase your n and outcomes reliability), one could follow-up this initial process with a more tedious review, should a positive outcome result from the initial evaluation. (My small groups analytic techniques are applicable to this.) We can then add to this specific sub-study with other follow-up questions, for example a review of medication use and compliance.
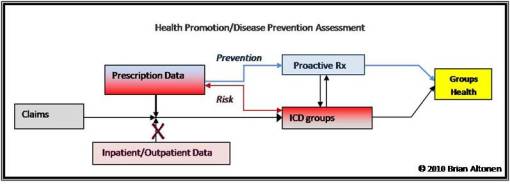
This particular methodology is why the above flowchart is used to produce a “Groups Health” review. By Groups Health I am referring to clusters of data that can be merged to avoid mistaken outcomes during the first pass through of the database under evaluation. These Groups may be defined by systems, as is the case for the Patient Compliance measure, or some other fashion that is either health- or cost-related may be used, one that is focused on a methodology and cause found to be comparable between the different components of that group for unexpected reasons. (I probably wouldn’t want to compare high cost in-house antibiotics use with high cost Mabs or interferons for example; but one moderate or high cost drug used to treat the same condition could be compared with the more moderately costing brand, for two or more auto-immune diseases).
The separation of Proactive from Reactive responses (defined as ‘Risk’ in the figure above) is done because of the significant differences between the approaches that would have to be taken regarding any interventions that could ensue. For a program with a small children population, we could merge the Childhood age (0-18) URI and Pharyngitis HEDIS measure results together, and then evaluate each separately should such a need arise due to a positive outcome of the study group as a whole. For a program with a small nutritionally-based chronic disease prevention/management program, a program with too few HTN, HL and NIDDM clients, a “Group” could be developed that combines these together to form statistically measurable numbers for the preliminary summary report, followed by a follow-up on each of these individual ICD/disease types. This group could be approached as a unit by way of some valuable intervention method, such as a clinical service-based change in scheduling and/or costs.
The following might be an example how these groups can be established, for a particular program type (Pregnancy of course always pertains to women).
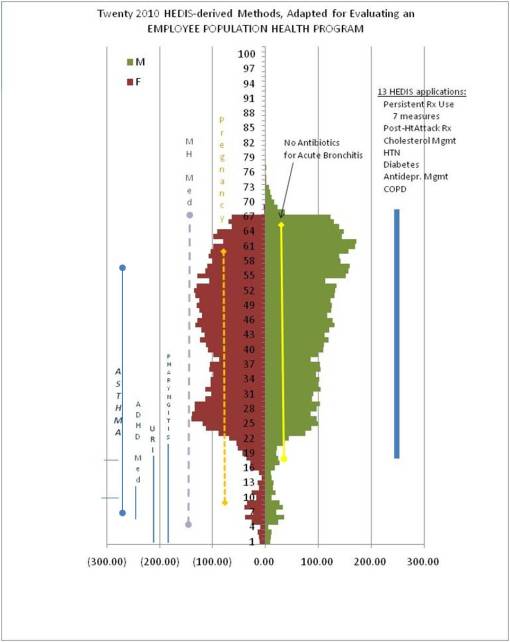
One of the features unique to this population pyramid is expected to be related to gender-age related differences. There are slightly more women in child-bearing years and therefore possibly a larger than expected dataset of individuals with an ICD or DSM for pregnancy, and the need for preventive care involving a larger number of children within the family setting. Whereas in the later years, women seem to be less employed than men, with men being employed slightly longer than women, this also suggests a possible need for review of certain conditions more common to individual who are 40 years of age and older, in particular chronic heart disease, diabetes, COPD, HTN, Alzheimers, etc.. To accomodate for these differences, one might also wish to add several other measures that are focused on other medical conditions important to this type of population. When such is the case, the addition of measures should be engaged in with the goal of adjusting for age and gender gaps noted in the population as a whole.
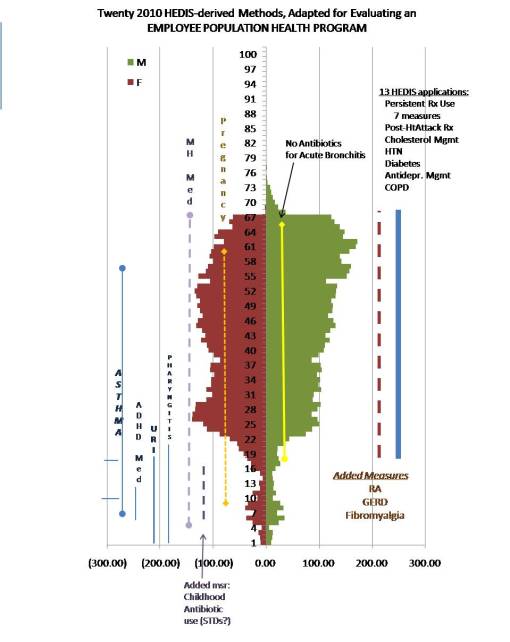
Once these adjustments in the research project are completed, a complete assessment could then begin. It is expected that a basic review of population health must consist of about 25 disease states and/or HEDIS topics, producing about 40 to 50 measures. In the following example of this type of evaluation, GERD was the focus of the reviewm but an equally important addition was fibromyalgia (which has a F:M ratio of approximately 4:1) in order to counter the focus on job-related chronic disease due to the higher numbers of men in this part of the population.
To improve the above project on population health even more, additional population health measures can now be added to produce a more robust study and subsequent presentation. A review of the prescription data could help define what additional topics may need to be added, based on unusually high rates for particular product types or classes. In another case, cost analysis could become an issue, resulting in the addition of an institutional or patient defined cost-related measure.
To complete this kind of population health review, since much of this review is based on prescriptional information from the National database, it might be of interest to CEOs and Managers to see a summary of these findings at product, script and systems levels. In addition, throughout this population health analysis project and in preparation of a final report, statistics on the utilization of prescription drug and non-prescription related items could be included in the body or addenda for the final report. Also, some of these reported statistics should have a larger population to be compared with such as the entire internal database reviewed for this project as a whole, or some national statistics (and as a final note, should some significant differences appear to exist with the national numbers, try to have ready an argument for this).