The Evolution of Natural Products
If we consider chemical complexity in the context of plant taxonomy and evolution, a number of basic rules can be applied to the development of chemical compounds and the new pathways formed for chemical synthesis in terms of evolutionary complexity. These rules are not unique to this study and have probably appeared in part, in some way shape or form in other writings on the evolution of plant chemicals, but these rules are important to understanding how taxonomy, time and chemical pathways relate to the overall chemistry of a plant and its ecology. Speaking in a humanistic sense, the evolutionary goal of a plant is to produce the most complex end products in the right places in the evolutionary tree and at the right times during the various evolutionary stages organisms go through.
When we try to interpret the effect of this evolutionary sequence on plant medicine development, we find a couple of basic changes take place in relation to the types of potential activities these compounds possess as medicines. The purpose of plant chemical evolution for much of its history is not to produce plant medicines, it is to produce protective agents and ecological protectants or enhancers. The new chemicals which plants produce have only recently in biological history become meaningful to mankind in general. There are some plants that are very dependent upon man for their survival, mostly those domesticated and naturally selected for by man, or genetically engineered by man, in order to improve upon their productivity and growth behaviors when raised in their natural settings, in their natural forms.
Elsewhere I define the four types of chemicals–4 levels of chemicals that are produced in plants. This retains the traditional primary natural products definition and redefines the “secondary natural products” term definition into three distinct parts–secondary natural products for environmental purposes, tertiary natural products for ecological purposes, and quaternary natural products for human ecological purposes. These four stages of chemical evolution and synthesis are also referred to as levels 1, 2, 3 and 4.
The following rules help to define the various components of phytochemical evolution related change. These steps define how a chemical can be changed, to indicate a new evolutionary test and application have been performed for the old and new products. These forms of chemical change are first listed together and then each is covered in more detail separately.
RULES
- Chemicals evolve from simple to complex in terms of structural appearance, pattern and make-up.
- Chemicals of a single class (chemical type such as saccharide/sugar, polyketide/fatty acid, polyterpene) usually evolve from small to large in terms of end-product molecular size.
- Chemicals tend to combine themselves in one or more ways so as to produce a one-to-many transformation (polymerization), with increasing amounts of branching; more specifically, they tend form larger and more complex three-dimensional molecules through polymerization or merging two or more nearly identical building blocks.
- Chemical pathways tend to diversify in their preparation and selection of building blocks, resulting in the diversification of the potential applications for these end products
- Chemical pathways tend to become increasingly more integrated as we ascend the evolutionary stepladder; highly advanced synthesis pathways often incorporate end-products produced by two or more completely different chemical pathways, so as to result in the evolution of a new (third) type of chemical pathway
- Throughout all these changes, the purpose or goal of these modifications are to produce an end product that adds to the natural selection process and survival traits a plant may have.
![]()
RULE 1.
Chemicals evolve from simple to complex in terms of structural appearance or pattern and make-up.
Saccharides. Sugars are the most classic example of this evolution noted in many pharmacognosy books. Sugars are defined as compounds with CH2O building blocks fashioned as H-(CHOH)n-H, with the end carbons often combined via an Oxygen to form a ring. In theory the number of carbons in this group is 3 or more, but typically we see 4C to 6C, with 3C compounds occasionally formed without cyclization. The two basic forms of moeities of sugars are 5C rings (pentoses) and 6C rings (hexoses). C7s and C8s are also sometimes noted in discussions of plant sugars.
Examples of the most basic sugars in plants are as follows:
Maltose and Apiose (4C rings) –> Fructose (penta-ring) and Glucose (hexa-ring) –> Rhamnose (deoxyhexa)
Sugars have modifications in their basic structure that are important to note as well. A sugar with an additional -OH group is a sugar alcohol. These alcohols are not as readily matabolized as sugars usually, and tend to by more hydrophilic. In the plant cell, they might serve as intermediaries in chemical paths, but they add to the plant’s cellular make-up or cytoplasm a little more hydrophilic ability. Whereas humans release sugar alcohols into the urine making them somewhat diuretic (very effective in the case of mannitol, which is so hydrophilic it can pull water from the edematous brain and reduce cerebral swelling in very short time), to the plant cell or tissue they aid in tissue construction and form and water retention.
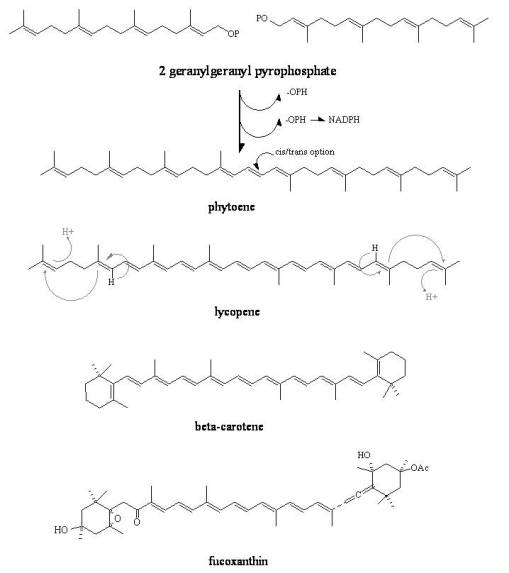
Carotenoids. A completely different group unrelated to sugars, carotenoids, is another example of this act of nature. The most basic carotenoid is a product of two building blocks merged via a pyrophosphate enzymatically driven reaction to produce phytoene, a simple precarotenoid chain with terpene (5C, iso, see later) shapes that are well delineated. A series of reactions then ensue producing the final structure of the carotenoid end product. Basic paths of synthesis will produce acyclic forms of this class of products, the most common of which currently is lycopene (the red in tomatoes). As these paths become more evolved, we see end-chain cyclization occur, which is still a fairly basic carotenoid forming process, followed by more complex reactions related to side group additions. In the example below, the fucoxanthin is actually a natural carotenoid product (fucoxanthin) found in brown algae; the saline environment facilitates ionization reactions like the formation of O-Acetyl linkages to the carotenoid base to form the brown pigments characteristic of brown algae. Ionic reactions like this are not unusual for algae residing in saltwater environments. In the Plantae, such a production is considered highly advanced, and would usually be related to a fairly biologically active substance such as a toxin or protectant. In the case of algae, the fucoxanthin provides the organism with the advantages needed to survive in salt-rich ocean water, it adds a polarity to the cytoplasm that balances out with the highly ionic ocean water medium.
Carotenoids:
![]()
RULE 2.
Chemicals of a single class (chemical type such as saccharide/sugar, polyketide/fatty acid, polyterpene) usually evolve from small to large in terms of end-product molecular size.
.
Small to large is like saying short-chain to long-chain, or oligomeric to polymeric. The general rule with increase in molecular size is that this is done in part by a plant as a result of a continued synthesis pathway. The plant has evolved a route for forming something, continues the production process one or two steps further than expected, resulting in a product that is of some advantage to survival. However, this fairly humanistic way of defining this process is somewhat simplistic. Plants do have a direct reason for producing very long polysaccharides: the molecule stores more energy for them, and may serve another purpose that is highly advantageous to the plant, such as increased hydrophilically-induced water-storage and holding capacity (for plants in exceptionally dry environments). It is important to note that the pathway to generating this product requires energy to start, perhaps more energy than is needed to continue, so the production line continues its process once it begins. Once a polymerization like process begins, it is easier to continue the metabolic process than to end it, only to have to expend energy later in order to reinitiate it.
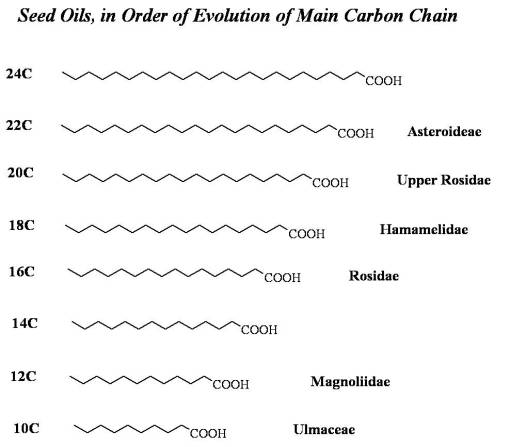
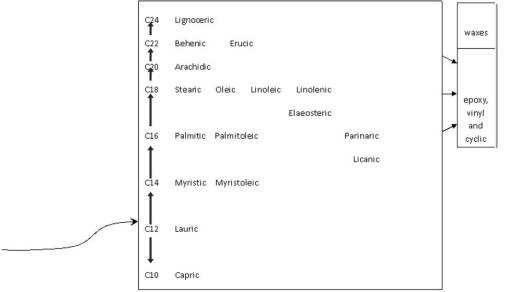
Fixed Oils. The chemistry of fixed oils is an excellent example of increased molecular size with evolution. Notice the progression of fixed oils from short chain (C12-C14) to very long chain (C22-C24). The names of subclasses, orders and families are provided to demonstrate an evolutionary process here. Fixed or seed oils are some of the best examples of this evolutionary process. The early evolving Magnoliidae of the tropics have short chain fatty acids, more than adequate in tropic settings. They are quick to produce and do not require too much merging of 3C to 2C generated precursors needed to generate the oil chain. The seed oils of the tropics are predominantly short chains (<16C chain usually). These oils behave well in a tropical environment, remaining liquid and less viscous in normal biological conditions. However, they are likely to oxidize, become acetylated or peroxidated forming ketones and the like. So with their small chain, the greatest risk of the short chain is its instability and likelihood of modification processes developing due to environmental exposure, a process possibly counter to survival.
For this reason, it benefits the plant to produce long chain fatty acids if the prevailing climate so allows, which we see taking place in families more adapted to temperate zone climates. This too can be seen in families that exist in regions ranging from temperate to subpolar environments. The coldest environments tend to make it beneficial to plants to produce these very different fatty acids. The cold climate oils are not only longer chained (usually long-chain=cold, medium chain-warm temperate evolution), they also tend towards double bond formation and main chain and side groups reactivity such as the production of vinyls (C-C triple bonds) or the production of -C-O-C- tricyclics with the -O- bridging the two Cs. Double bond formation and the desaturation of a C-C main chain produce very unstable structures in warm to hot environments. An excellant example of this environmentally-selected processed in demonstrate by the Hamamaelidae (Catkin-bearing trees that produce nuts and other sizeable seed-like structures.) This grtoup evolved in and has members that are well adapted to colder climates, and so most of its members are rich in long-chain polyunsaturated fatty acids. [Only C18 is depicted below; these unsaturated Fatty Acids are not shown below.]
Terpenoids. We have already seen an isoprenoids/terpenes association with carotenoids. In this one to many scenario, we are talking about hemiterpenes increasing in size to produce monoterpenes to sesquiterpenes. The building block is a C5 structure (the terpene or isoprene), generated by the merging of two C3 building blocks, with the loss of one carbon. This is a very common path in plant evolution and has two major evolutionary routes of development. Both may have evolved from the same synthesis pathway as a base. The Pinidae/Coniferae evolved their terpenoid resins from this, which are very different from the resins and lighter polyterpenes produce in the flowering plants, for the most part. Pinidae polyterpenes are that thick viscous resin found on the conifers, an evolutionary advantage that tends to work well in both cold and warm climates. The polyterpene path is Pinidae produced its characteristic sticky resins in the Northern hemisphere, and less sticky, more brittle when dried resins when developed in the southern hemispheres. Either way, these resins and their demonstration by the plant for whatever evolutionary reason, are very sticky and tacky, unlike most flowering plant displays of terpenoid products.
C6 (hemi) –> C12 (mono) –> C18 (sesqui) –> C24(di) –> C30 (sester) –> C36 (triterpenes) –> Cn (latex)
Flowering plants utilize a more oxygen-rich terpenation process, resulting in the formation of mostly oxygen-rich monoterpenes to triterpenes, and few exceptionally long polyterpenes–namely the latex bases like those related to rubber, gutta and chicle. These oxygens make the terpenoid more reactive to other compounds, and more sensitive to temperature, light and other energy-related events (i.e. the production of free radicals by oxygen rich oligoterpenes due to light exposure). When we look at the development of terpene products in plants, we see long chains tending to form more in hotter climates. This is due to the rapid loss of terpenoids, especially highly volatile light molecular weight molecules, in these hotter climate settings. For this reason, resins are more developed and more unique in warmer climate settings, with those formed by flowering plants in desert settings serving as excellent examples of the variety of these types of end products that can be developed. Warm to hot climate resins tend to be sequiterpene (C15) in size, having lost a lot of their more volatile hemi and monoterpenes. A single genus with species in two climatic settings, or a family with closely related genera in two different climate settings, will produce essential oil like products in one climate, and resin-like products in the other. This is especially the case for umbel family members, for example to Ferula species of the arid setting (resinoid rich like asafoetida and sumbula) versus the fennel and allies of the temperate Europe to Mediterranean climate settings, versus the cooler temperate region members like parsley and dill.
![]()
RULE 3.
Chemicals tend to combine themselves in one or more ways so as to produce a one-to-many transformation, with increasing amounts of branching; more specifically, they tend form larger and more complex three-dimensional molecules through polymerization, or merging two or more nearly identical building blocks.
Examples of chemicals that follow this rule:
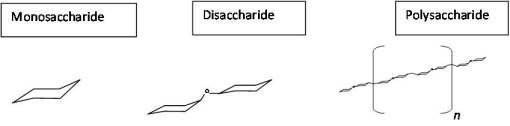
- single chained polysaccharide to branched chain polysaccharide. Process: monosaccharides to disaccharides . . . . to polysacccharides (starch and mucopolysaccharides) to branched chain polysaccharides.
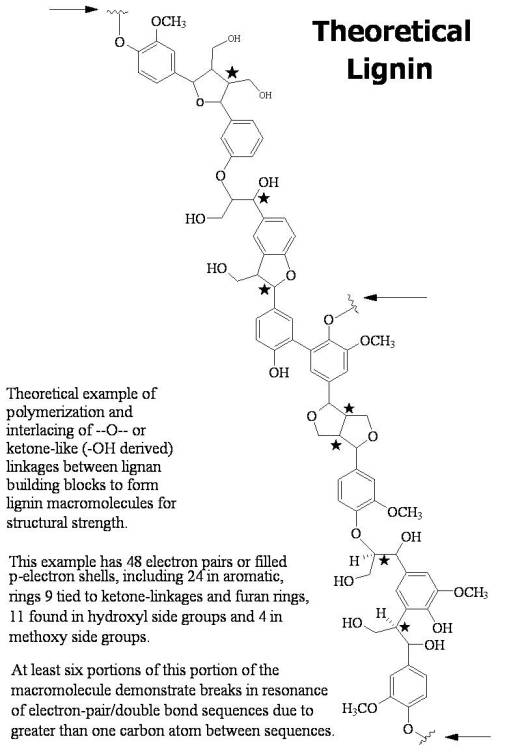
- Lignin, produced by merging pre-lignin building blocks called lignans. The following is a theoretical form lignans develop to help strengthen woody-plant parts with lignin:
In general, branching provides an organism with a much more secure way of storing large amounts of chemical energy. However, the three-dimensionality of branching also makes for chemical products designed to serve primarily a structural purpose. By producing a fairly stable matrix of long chain molecules, many interwoven and bearing H…H bonding (hydrogen bonding) features as well, what results is the production of a stable chemical environment. If we look at this as just a structural adaptation that a species can go through, the best example of such an important effect is seen with mucopolysaccharides. These long chain polysaccharides, with side chains, polarized side groups and H…H (hydrogen) bonding features make for a system known as a colloid, in which the system in theory has numerous independent components that due to their unique blending results in a very stable seemingly solid structure. Colloids are responsible for producing cytoplasms that help to maintain the structure of the organism at the cellular level, in which the system is fluid like in behavior but still stable in structure. Colloids are also responsible for producing end products that appear and behave very much like solid structures at some macroscopic level. By adding just the right amounts of ingredients capable of binding the large chain-like molecules together, for example the process associated with the formation of dough from flour and water, these branching molecules become an essential component to the recipe required to stabilize a final structure. In this way fixed oils interweave with polysaccharides, polyterpenes and ionic phenols to help form gums and resins, and at the extreme end of this process, lignin and cellulose go on to form very stable wood.
![]()
RULE 4.
Chemical pathways tend to diversify in their preparation and selection of building blocks, resulting in diversification of potential applications and uses for these end products.
A single building block is one avenue plants take to polymerization. The use of different kinds of building blocks, especially in some non-random fashion, is a great deal more evolved. For example, the production of a starch containing very different saccharides can be such a process. The addition of odd-number verbascose and rhamnose-derived building blocks in order to generate starch-like muciloid protectants around legume beans in a pod is an excellent example of this. These muciloid boundaries are slightly more polar, hydrophilic, and are capable of adding water to an environment once the bean is ready to propagate. This mucilage also contains some feeding deterrents such as bitter tasting compounds and tannins. This highly polar and often colorful mucilage outside the seed varies greatly with the blandish-looking polysaccharide within the bean, a slightly modified polysaccharide more like the typical starch than a seed-protecting mucilage. The most common examples of these in food manufacturing are guar gum or locust bean gum.
We can also apply this rule to the fixed oils path. Fixed oils are used to produce waxes and other complex hydrocarbons, usually to serve as some form of physical protectant. In general, fixed oils evolve along the following type of path:
- saturated short chain [i.e. Magnoliidae, short chain, saturated]
- to unsaturated medium [much of the Eudicots]
- to long chain, and polyunsaturated long chain, [two branches: at the primitive level–Hamamaelidae PUFA long chains, otherwise advanced Eudicots levels, esp. Asteridae-Asterales, and advanced Umbels]
- to EPA or PG-like polyunsaturated, or cyclic (end- or mid-chain cyclization), advanced eudicots (see Dilleniidae hydnocarpic acid and allies, Malvaceae seed oils with cyclization)
- to vinylization (double desaturation, forming triple bonds), esp. seen in Asteridae-Asteracles-Asteraceae.
- to waxes (esterification reaction based polymerization process–two fixed oils merged at identical ends of the chain, R–COOH and R’–COOH forms R–COOC–R’)
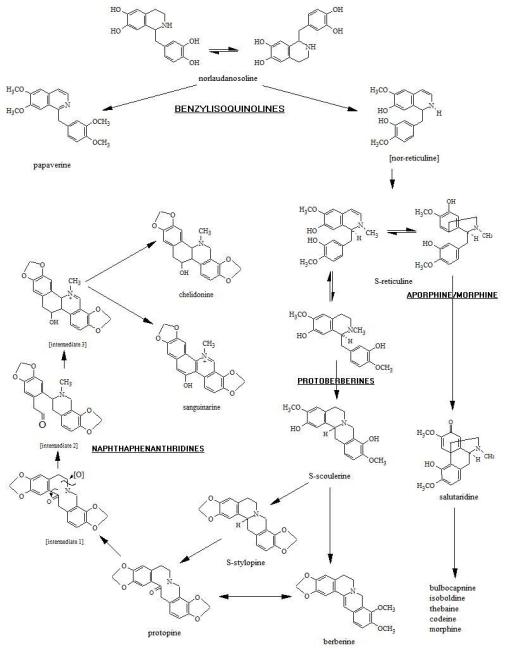
When we apply this rule to the formation of a broad class of alkaloids like the benzylisoquinolines (BIQs) we find there to be very good examples of this modification of chemical form to more complex structures. For the BIQs, we see ring structures that progress from 2+1 (benzylisoquinoline) to 4 (protoberberine) to 5+ (naphthphenanthrene) cyclicity. The increases in double bond aromatization in part lead to a “darkening” of the resulting color of these alkaloids on the rainbow (red colors are longest molecules with most rings and most alternating double bonds; yellow have less of these–the half-sized alkaloid break down product hydrastinine, a product of the yellow alkaloid hydrastine in goldenseal, is sky blue under SW UV).
A continuation of this process as part of the terpene related pathway demonstrates steroid formation, with unique side group add-ons produced in the most complex steroidal (triterpenoid) products. Comparing the basic steroid skeleton and two highly specialized phytosterols, below, we see two very different pathways of development. Brassinolide is hormonal and inhibits growth of competition. Solasodine is alkaloidal and selective toxic. Another interesting example of this is the formation of reversed triterpenes (rTTs) in the Cucurbitaceae and allies within the plant evolution tree. These rTT’s tend to be highly toxic and can often be effective cancer remedies. This conversion of steroids transforms a basic phytosterol from a primary and secondary product (using my definition of these groupings), into tertiary (environmental-ecological) products (feeding deterrents and such, such as the butterfly weed or solanaceae alkaloidal cardiotoxic sterols), and from this class into quaternary products (used predominantly by man, such as the rTTs for cancer drugs).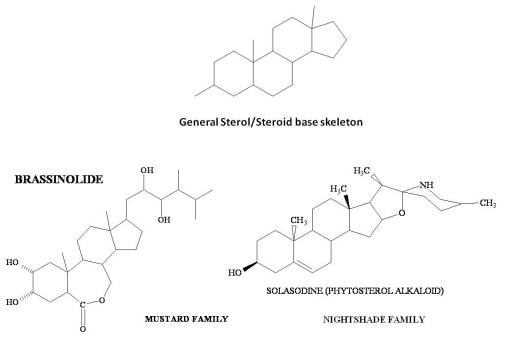
Reversed Triterpenoids:
![]()
RULE 5.
Chemical pathways tend to become increasingly more integrated as we ascend the evolutionary stepladder; highly advanced synthesis pathways often incorporate end-products produced by two or more completely different chemical pathways, so as to result in the evolution of a new (third) type of chemical pathway.
The following examples are pretty much self-explanatory. The merging of various unique paths always results in new products that are very important to a species evolution. This process does not usually result in structurally adaptive features although it can, like the merging of tannins, lignin and cellulose pathway products to form wood. It usually results in some highly evolved groups of compounds, the most impressive of which are the alkaloids produced by the most evolved subclass of doctors–the Asteridae–Asclepiadaceae and its neighboring Rubiaceae and allies–the sources for the largest collection of the most toxic alkaloids in the Plant Kingdom.
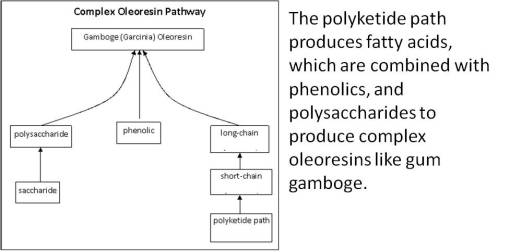
The advantages this process provides for plants are obvious. By mixing a resinoid path with a highly evolved polysaccharide path and/or a moderately to highly evolved fatty acid path, the end products that are generated (i.e. Garcinia gamboge oleoglucoresin) serve a very specialized purpose, to the plant, and ultimately man.
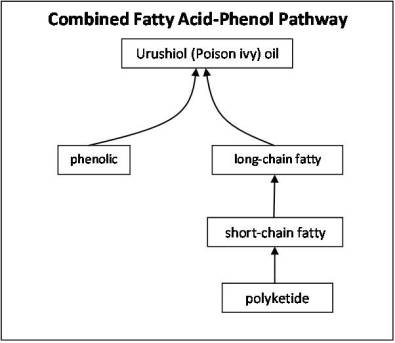
Likewise the toxic oil in Rhus species is formed by merging products from the fatty acids synthesis paths with phenolics. The fatty acid enables the toxin to penetrate and exert its effect through the skin. The phenolic is the immunogen, the results of which are the unique reaction that causing blister along with the spread of this oil physically in various directions due to white blood cell activity (macrophages attempt to surround the oil, causing it to split into two drops, which are again attacked and subsequently split, etc.).
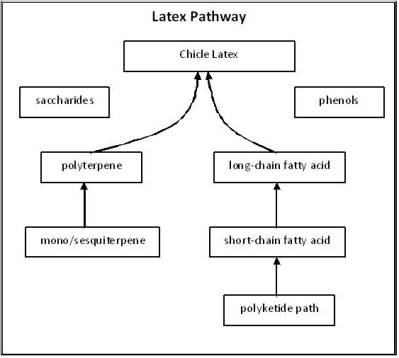
One of the most unique highly complex phytochemical paths results in the production of latex. Produced by a unique cell called a laticifer, this latex has an exceptionally long chain polyterpene as its base. With the evolution of more advanced versions of latex, we find significantly modified side groups on these terpenes and the nature of the bonds that are formed result in unusual physical chemistry effects. For example, the difference between normal latex rubber and chicle is the presence of more polarity and modified bonds in the “terpene links” forming the chemical chain of polyterpenes. This structural and side group difference makes chicle less elastic and “rigid” (able to return to form) than rubber, but more viable for other unusual uses (chewing gum). Even more modifications of the fairly consistent polymer of rubber results it the very inelastic end product gutta percha. These simple chemical differences in polyterpene chains are combined with other latex chemicals to product the final plant-produced raw latex end-product. This end product is often made fungal-proof and bacteria-proof naturally be the plant due to the phenolics that are added. It is less likely to be consumed due to distasteful tannins. The natural oils in latex enable it to be modified and turned into a suspension. A latex may be made more hydrophilic by the plant by adding saccharides, thereby controlling the ability of a plant to best meet its specific viscosity needs for the time. As mentioned previously, the addition of alkaloidal steroids to a latex can make the latex of certain species more toxic, like those of the Apocynaceae.
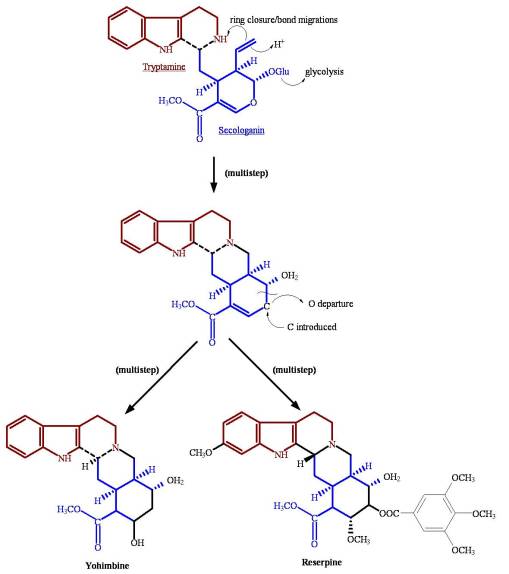
The seco-loganin alkaloids path of the more complex Asteridae is perhaps one of the more impressive examples of the complex integration of multiple pathway. This is especially true for Rubiales members. The seco-loganin base derived from the third tier in the monoterpene to iridoid path is then diverted into another pathway of a completely different physiological origin. The alkaloidal (N-containing) path produces complex chemicals of an alkaloidal nature using a path already well established by various parts of the plant kingdom. When this alkaloidal pathway is merged with the seco-loganin path, the result is a uniquely varied polycyclic alkaloid group with major differences in selective toxicity defined by the overall three-dimensionality of the end product. Two seemingly similar alkaloids can have vastly different forms of selective toxicity due to the 3-D change and placement of the N-groups. Many of the most toxic plant alklaoids are produced in this fashion. (See the last example on this page.)
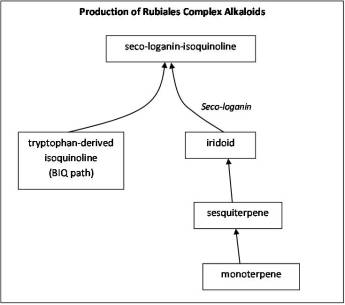
RULE 6.
Throughout all these processes, the purpose or goal of these modifications is to produce an end product that adds to the specific natural selection values and traits that a plant may have.
This is an example of classical Darwinist thinking. The change in color of a plant petal to better attract pollinators is example of an evolution-based, genetically-related chemical change. The change in the toxicity of a seed due to the modification of a common amino acid into a highly toxic non-protein amino acid, thereby protecting the seed, is an other example.
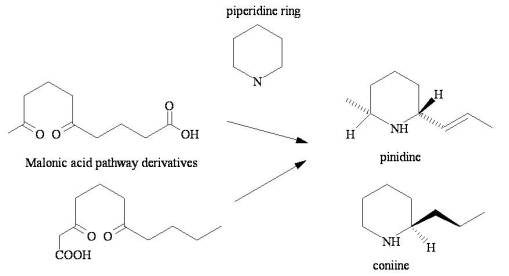
Even the simplest of chemical reactions can result in toxic substances that are of evolutionary advantage. One of the simplest examples relates to one of the older taxonomic groups–the Coniferae. The antifeedant Pinidine from pine is a side product of a C2 (from pyruvic acid) path that normally might otherwise be used to produce other fairly complex chemicals, but in this case stops after developing a C10 derivative. Another simple highly toxic end-product, Coniine (the toxin of water hemlock) is a pseudo-alkaloid produced in much the same way. The Nitrogen for each is derived from atypical, usually non-ring sources, and is added to the chemical by replacing the two oxygens in the two -C=O side groups, thereby closing a ring that has no double bonds.
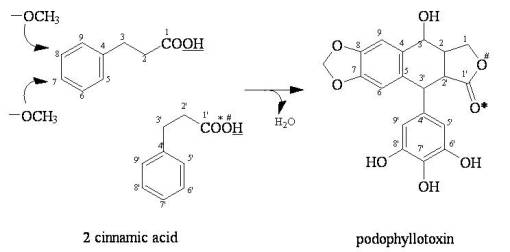
Example 2. In this next example, the simple joining of two of the same compounds produces a highly toxic cancer drug compound podophyllin. Like the above simple alkaloid process, this too has examples that exist in both fairly primitive divisions (Pinidae-Pinales-Calocedrus inner bark forms epipodophyllotoxin) and a herbaceae member of the paleotypic Magnoliidae-Ranunculales (Ranales) Order-Podophyllum peltatum. This seeming complex multi-ring structure is fairly easy to make.
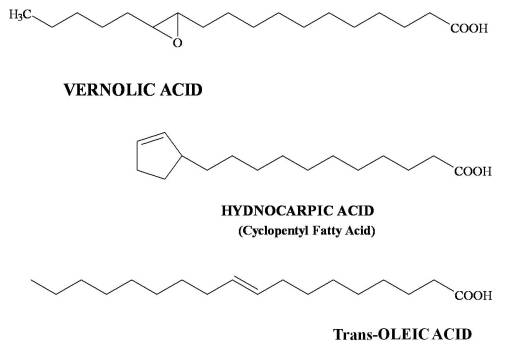
Example 3. The modification of the basic C-C chain in fatty acids not only changes the nutritional value of an oil but also increases its toxicity. One of the most important characteristics of a fatty acid is in hydrophobic nature. This is responsible for its ability to fairly rapidly enter biological systems thr, and ability to employ end products as medicines. In this manner, oleic acid is converted to various forms and/or new fatty acids due to cyclization. The more complex the transformation, the more likely toxicity can occur by introducing the product into normal biological system through semipermeable surfaces like the oral and gastrointestinal mucosae. But the increased complexity of a fatty acid also goes hand in hand with an evolution of selective toxicity due to this change. The simplest example of this is the modification of the cis-oleic acid into less common (but still natural) trans-oleic acid, which works differently along the typical paths in the body . The -COC- tricyclic formation in Vernolic acid demonstrates toxicity due to the free-radicalization reactions it can produce. Hydnocarpic acid and other pentacyclic fatty acids of varying chain length in the Dilleniidae are selectively toxic to microorganisms, and have an effect that can effectively penetrate cell membranes, even with fungal cells (a cause for one form of leprosy), due to the fatty acid base of this selective toxin.
Example 4. Seco-loganin (iridoid) pathway combined with alkaloid pathway produces most toxic plant alkaloids. The chemistry and very different selective toxicities produced by these two otherwise very similar chemicals require no explanation.