REMOTE SENSING
Applications
Disease Ecology
Most of my remote sensing experience pertains to disease ecology research. In particular, the applications of remote sensing analysis to host-vector borne disease ecology such as for west nile has been the focus of this work for most of the past 7 years. Prior to this, I researched the use of remote sensing to review landuse changes over fairly long periods of time, during which time my emphasis on plant ecology and vegetation domains helped to prepare me for the projects about to be developed for west nile surveillance.
My first project of vegetation domain analysis was actually an opportunity that arose as part of a Central American research project devoted to natural resources industries established by indigenous groups in and around Michoacan, Mexico. At the time, I was reviewing the techniques and methods used with remote sensing to identify plants from a considerable distance due to chemical content. This chemical content is the primary reason plant reflectance patterns often have unique features that help to differentiate them from other ecological settings. The best evidence for this related to the reasons why soil and geologic surfaces appear the way they do in multiband RS images.
My argument in favor of engaging in this use of remote sensing to better understand plant ecology was the fact that RS was already used to identify geochemical features, such as bedrock, mineral richness and soil inorganic materials content. This information is typically used to identify potentially new oil fields and mining sites. The differences between organic and inorganic compounds are pretty substantial though, leading to slightly different methods being relied upon to identify one feature versus the other.
Whereas the mineral make up of terrain, although variable, produces predictable patterns in most cases, plant chemistry varies considerably more from place to place and the sensed clues that we get from RS about this chemistry are most helpful in making identifications that are otherwise not well-presented by a visual identification of the plant. More appropriately, when we look at chemicals in plants for remote sensing purposes, we are not just reviewing a very small part of the organism, but also its potential impact of the environment and vice versa.
If we place a plant and a piece of mineral side-by-side in front of a remote sensing tool, what we would find is that the mineral for the most part is similar throughout in terms of chemical make-up and some rules can be developed and followed to make an identification. With plants, identification depends on the time of the year, the amount of water the plant recently had, whether or not it has matured or is still a seedling, whether or not it has received all of its necessary nutrients, whether or not it has received a toxin from the environment or soil, whether or not it is flowering, whether or not it is fruiting, whether or not it is magnesium-deprived, etc. etc.
Regardless of whether or not the remote sensing (RS) process with plants takes place proximally (close-by) or from far away, there are three major features of plants that are unique to them as parts of the local ecology and play important roles in this investigative process.
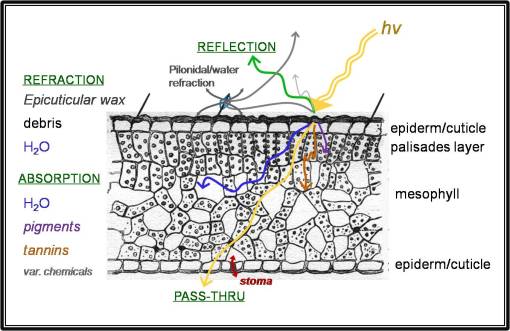
First, there are the effects of pigments in plants and how these pigments interact with incoming solar energy (think of these as visible light, ultraviolet and infrared, although there is a great deal more to this than just these three groups of solar-derived light energy). Second there are the effects of water content of plant cells and tissues on the remote sensing process. Third, there are the effects of miscellaneous chemicals in plants and how all of these chemicals interact with light when the plant structures are interpreted as tissue and organ, or organism-based systems.
The most common group of plant pigments reviewed with regard to remote sensing are the chlorophylls (green pigments), but there are other substitutes for chlorophyll like the xanthophylls (carotenoid-types engaged in light energy absorption), and anthocyanins (usually associated with the purple or reddish color of photosensitive structures). Anthocyanins are a type of flavonoid chemical, meaning that other flavonoids can impact plant tissue interactions with light as well. Flavonoids that are capable of adding color to a plant can be yellow to orange in color (these are most often associated with flower petals), blue, red, deep purple, etc.; essentially this group of compounds represents the complete rainbow of possible visible colours in the plant, even though this effect is hard to detect and differentiate from much larger effects seen by the major photosynthetic pigments.
Tannins are the next group of compounds that can impact plant color and remote sensing readings. Typically these are high energy absorbers that are fairly stable. We typically associate tannins with a dark brown to blackish color, but small tannins are usually colorless, yet still capable of absorbing certain energy forms. (Another group of “tannins”, the flavo-tannins, essentially fit in with the flavonoid groups discussion.)
Likewise, another chemical group capable of absorbing energy is the coumarins, especially furocoumarins and pyranocoumarins. Neither of these two sets of compounds are considered much in RS analyses due to the minimal effects so far noted and/or discussed by many RS specialists in plant chemistry. Coumarins tend to absorb at the shorter wavelengths or the ultraviolet (esp. short wave) range. The same can be said for other obscure and infrequently considered chemicals with potential for impacting RS data, like the highly colorful benzylisoquinoline alkaloids and their derivatives, the short wave sensitive iridoids, the possible absorptive effects of certain lignin-based chemical products, and the highly photoreactive and photosensitive quinones, such as the anthraquinones and dianthrones.
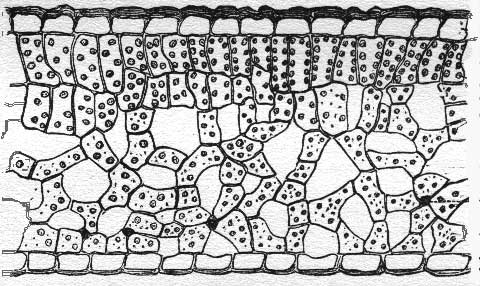
For plants, the concentrations of these chemicals responsible for producing the results we see with remote sensing is much less than the concentrations noted for specific chemical groups or types in minerals or rocks. These chemicals in plants may be distributed equally across the specimen or sequestered in one specific region. In some cases, the chemicals sensed in plants also have a semi-crystalline nature that has to present itself perfectly to the sun/sensing tool to be modified, become absorptive, and/or become detectable. This is especially the case for chlorophyll detection in plants, since this chemical resides in the chloroplast in association with a fairly sophisticated membrane and protein layout, arranged in such a way that it works much like a photosensor on a roof (e.g. a solar energy unit). For chlorophyll, the position of the molecule means everything when it comes to serving as an efficient energy gathering tool or not.
Looking at plant-related remote sensing in a much broader fashion, like at the plant structure-related level (the leaf, the bark, the cambial layer), we have a few more completely different set of problems to deal with. Due to the resolution of most common forms of remote sensing out there, we usually do not observe the plant from a distance and obtain information back from any other type of material residing nearby. We ususally obtain raster data with a resolution larger than a plant, with information obtained as well from neighboring plants, plants growing beneath the larger plant that is observed, and as well as dead plants parts and the actually soil, minerals and water lying in close proximity.
The next factor in dealing with plant-related remote sensing is the water content. As a living organism a plant can be as much as 90 to 95% water, with much of that water residing within cell membranes and in the mesophyll underlying the epiderm of the leaf or stem. If the plant is adapted to water-restriction related needs, then this problem is even mroe a problem since water can comprise a significant amount of the organism being viewed. With remote sensing, this water provides a sort of background noise that has to be filtered out in order to determine what else is really there in the organism.
Third, plants produce some of their own chemicals from scratch, obtain chemicals from the environment, and at times produce new types of chemicals as a result of environmental stress. This means that in some cases, it is easier to find a plant in stress, than it is to find a healthy plant. In terms of large scale remote sensing raster cell applications, these features are simply blended in with everything else found, and so may be completely lost in the analysis of the data.
Finally, chemicals is plants have inferior and superior qualities that can be attached to them. We can differentiate some features much more easily than others. With broad-band spectral imagery like that associated with Landsat and SPOT, we learn a limited amount of information that can be tied to large area plant features. With this limited information, we are able to use supervised and non-supervised classification methods to make an intelligent guess about what a particular remote sensing finding could mean.
For this last reason, remote sensing often requires field visits to verify the preliminary outcomes received after evaluating your images. With the greater resolution that is being offered by new companies, this becomes less the case, since often many plants can be indetified visually by air, without need to study their chemical content. Nevertheless, it helps to understand the chemical make-up of plants in order to better understanding other things that are happening in the immediate environmental setting. For this reason, I did an analysis of AVHRR data to determine if woukld be of any use and application to various disease monitoring and ecological research techniques.
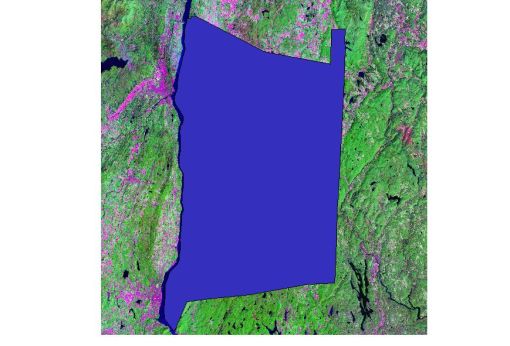
The following image depicts the county of Dutchess from the air (comparing it to a map, would you be able to see its borders?)
To define the county’s actual borders, it helps to put the landforms into perspective. The southern tip of the county, on the Hudson River, is the little “nose-like” form pointing down; this is the result of a mountain range stretching from eastnortheast to westsouthwest. The eastern border of the county is geographically marked by a narrow range of mountains near Connecticut and just east of a large highway known as Route 22.
Landsat images are produced using a different series of longitude-latitude lines, with positions dictated by a fairly complex math equation. When using Landsats to perform you work, they have usually been adjusted (the more adjustments you need, the more they cost) so that the roads on the image approximate where they are routinely placed in a GIS. However, at times it helps to develop you own modified GIS datasets to overlay on these maps in order to engage in a combined remote sensing-GIS method of presentation or review.
The following depicts the county of Dutchess overlaying the Landsat image.
To make use of these rastor images for analysis, it is a general rule that you either use the entire image provided for you or you cut it down in size to speed up the processing, making sure you include two or three more raster cell rows or columns beyond the border of the defined research area in the image. These extra cells make it so you can perform neighbor related spatial analysis and have an actual neighbor to be reviewed or compared with. In the case of the use of an averaging equation to modify you image for example, you want to avoid averaging the exact edge of the county with zero data; the end effect of doing this is greatly modifying or falsely displaying the edge of the image.
With the exception of some Hyde park studies, most of the analyses performed pertained to areas in the core of the county. So, the dark blue image noted above was used as a “cookie-cutter” to extract the needed Landsat data and NDVI obtained from the original landsat image set obtained. Most of the analysis performed for these next projects made use of IDRIS32 to perform the calculations and tally up the results.
The following are some of these projects involving West Nile.
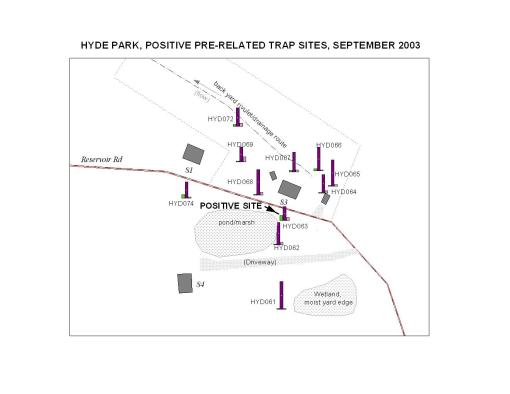
A Mid-County Positive Site
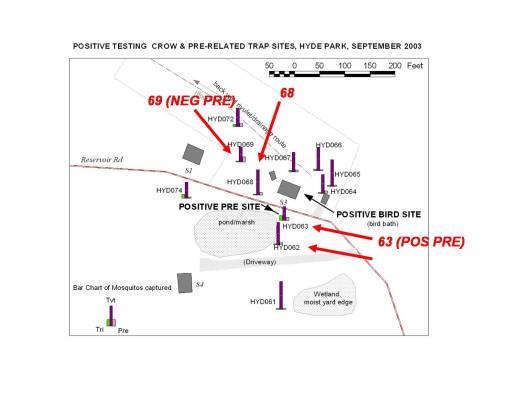
In one particular part of the county, a positive Crow species was identified followed by a report of another dead crow across the woodlands in another residential area; the individual who filed the second report stated that a person in his house just a few days earlier had just come down with west nile. As a result of these calls, attempts were made to do an ecological study on the area to determine whether of not it was possible for west nile to develop in that area based upon the physiographic and ecologic features.
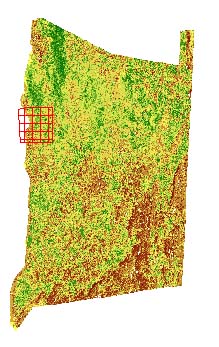
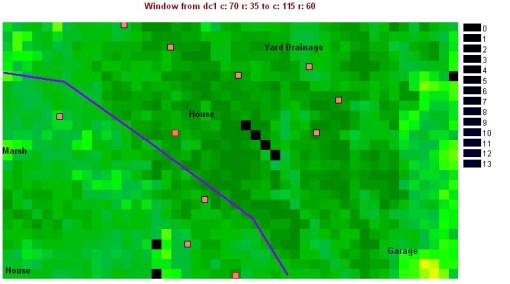
Using Idrisi, the research area was first located on the Normalized Difference Vegetation Index (NDVI) map.
A large number of traps were set on the immediate property to determine if the crow could have been infected locally. Of course, this was more than a week since the crow was reported and the possibility that any positive testing vector could remain in this setting was fairly low. For this reason, aside from concentrating on trying to locate a nearby source for the west nile infected mosquitoes, another ecological study was performed, based on the landowner’s claims that the crow may have come from any of several directions across his nearby lawn and field (the site was originally used as an orchard and the caller’s home its farmhouse).
Numerous small waterbodies were identified in the immediate area, requiring that traps be set in numerous places including the roadway and a neighbor’s yard edge, two sides of which consisted of small water bodies (a round glacial ice pond about 25 feet across with significant cover and canopy light penetration in the appropriate range, and a swamp with significant cover and canopy light penetration in the appropriate range). There was also a seasonal creek that traverses the back yard area of the house that reported the dead crow. Throughout the yard were placed a variety of fruit bearing apple trees in combination with much large oaks scattered across the back yard. The far back edge of the yard had a grove of evergreens about 40 feet in height and which provided a very well-shaded retreat site useful for nocturnal species during the daytime. A small area map was produced for the entire area, including two households, the roadway, driveway, waterbodies and any other major homestead-related landuse features.
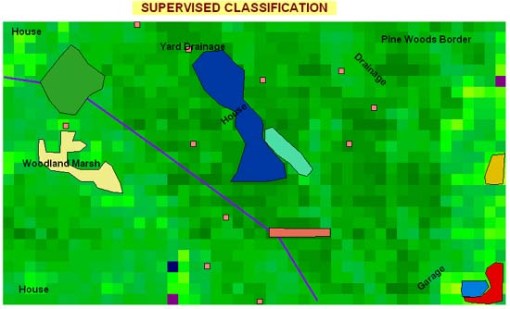
The first project for this research setting began with a supervised classification in order to define the specific uses of areas for which uses were identified based on field visits. Specific types of areas to pay particular attention to were located and then the Idrisi program told where these sites were. Idrisi then uses this information to reclassifiy the rest of the image in the research area.
This was used to resample and reclassify the image so as to define the particular foliage pattern for the region.
The following key defines the different areas identified by their color changes.
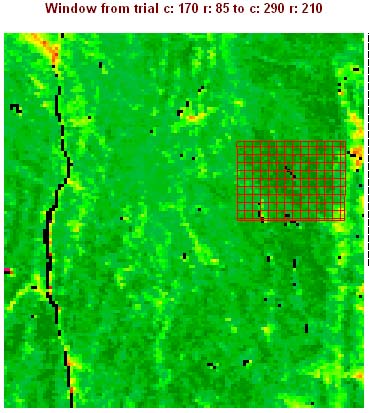
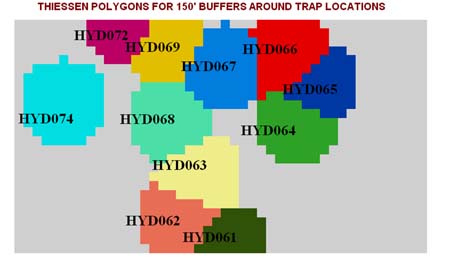
In this rectangular research area, each trap set covers a specific area of the land surface. Through the use of a Theissen Polygon technique, we can assign each trap that is set a particular portion of the property that it covers. In this stage of the analysis, fuzzy logic is not being employed because we are not focusing on the mosquito vector, which is capable of selecting whatever trap it wishes to fly towards. Instead, we are looking at the specific area around the trap and how it ecologically fits in with this scene as a whole. It is meant to answer the question: What overall surface (phytoecological) features may be used to define why the mosquitoes fly the way they do?
Next we assigned circular areas to each of the traps set, because some areas of the Thiessen polygons are too far away from the traps to be considered relevant.
To fill in the missing gaps of information on the trap setting activities, we produce the same sorts of maps for the previous images.
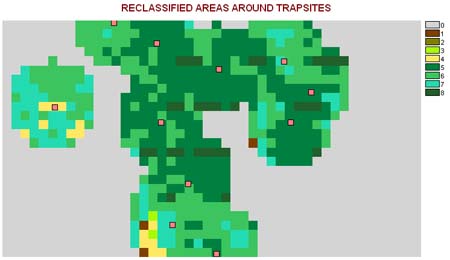
This information is then used to define the best gridcells for finding west nile mosquitoes, or to set the traps. In the following image, these areas are assigned a risk value by using a standard reclass method, ranking the results on a scale from 0 through 8, with 8 indicating highest risk (8 is used instead of 10 due to bytes or 2^n data storage capacity for each cell in this type of grid and the nature of the IDRISI programming tool). The following application of this reclass of a reclass can then be reviewed in the field to determine what outstanding ecological features defined these highest risk areas. This in turn tells one where to concentrate for prevention activities in terms of integrative pesticide management practice, and can be used to inform the land owner and community as a whole as to how to better prevent high risk areas from developing.
In the following image, the above image legend was redefined to make the common area closest to the home and immediate yard settings and walk areas stand out in blue and the highest risk small area gridcells located immediately adjacent to this high risk/high chances for exposure area area in pink. The second highest risk areas depicted in yellow represent wetlands type land surfaces (fairly moist, but not with permanent water bodies). Adjacent to these (see western-most theissen buffer) is the next high risk area (green gridcells = risk level of 6) with considerably high water content–the edges of which possible define an important breeding site, although not necessarily an exposure site due to insufficient canopy protection.
The Biodiversity of Plants and Vector Ecology
How we should apply Landsat and NDVI images to evaluations of the ecology of west nile is to use these images to determine how much of a role each particular type of vegetation region plays on the disease ecology pattern. Identified by Idrisi, these sites are then evaluated to see how much of a role they play in defining the trapping results and the absence or presence of positive testing west nile hosts of vectors (numbers are site identifiers).
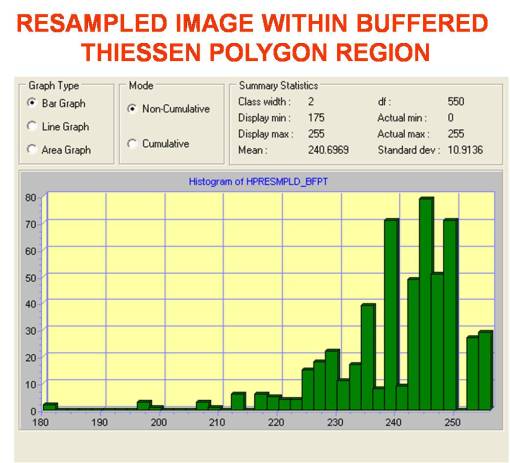
Referring back to the Thiessen polygon approach that this part of the project began with, we can evaluate the raster cell features statistically to determine if there is one or more particular cell type(s) (underlying ecotypes) represented by each cell that stand out in west nile areas.
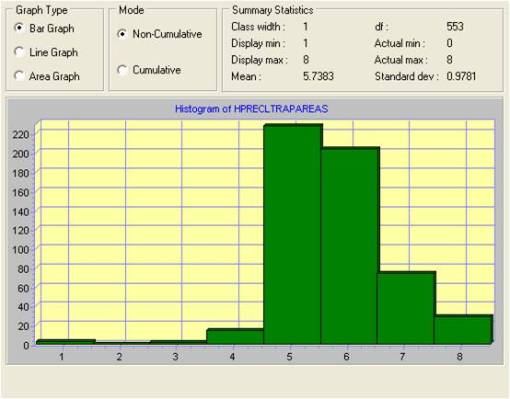
Several possible high frequency cell types can be seen on the histogram for this area. Since there are several ways we can evaluate ecological settings regarding plant canopy cover using GIS/RS tools, we can do some fairly basic evaluations of the are to determine if any traditional biodiversity related features stand out based on the formulas used to evaluate biodiversity in ecological settings. Species richness is a variation on biodiversity that tells us whether or not areas with large numbers of species (based again on how grid cell values are interpreted by the program), not taking into account as much the density of these particular species in an additive fashion spatially (using a matching nearest neighbor approach for example). With this reasoning, the following standard types of images were produced for the multiple trapping area.
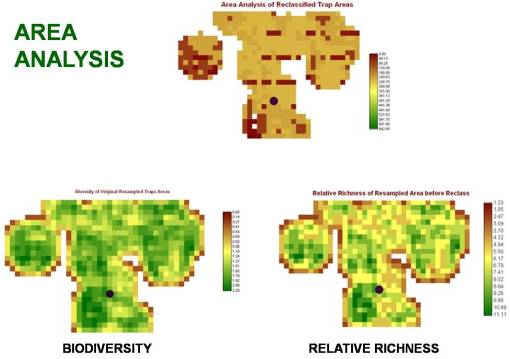
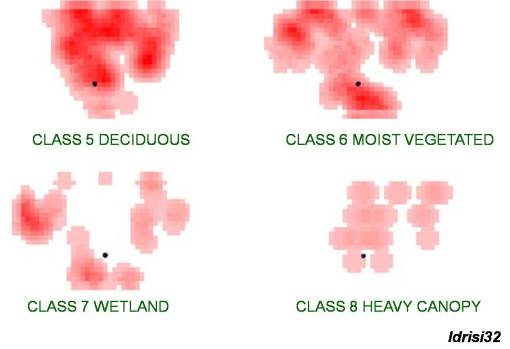
Three evaluations of the trap sites in a positive testing West Nile area. (The point represents the positive crow case.)
One of the more useful Idrisi tools applicable to west nile phytoecology analysis is a vegetation reclass tool available for evaluating NDVI imagery with the Idrisi GIS-RS software. This tool uses a formula to analyze surface features based on cell color (wavelength data) and then applies a cluster analysis formula to define areas with particular features (these areas are defined by the supervised sampling cell values described earlier), such as the driveway area, the house, the yard/wet grassy region area, a wetlands area, a large tree canopy area, a roadside area, etc. Applying this methodology, the research area defined by the Thiessen Polygons and 1 or 2 cell buffers placed around trapsites is assigned the following vegetation-based reclass types.
NOTE: Several other types of areas that are not relevant to the vector/disease ecology are not displayed here, such as a garage/homestead-related area.
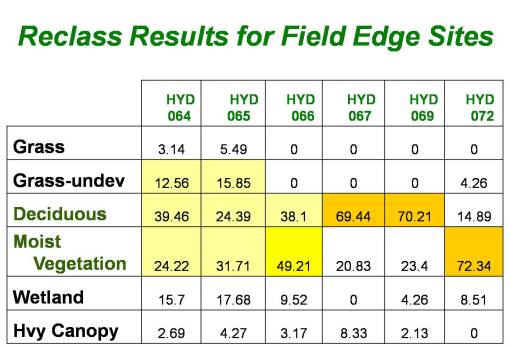
The amount of association between each of the Idrisi-defined important vegetation regions and the west nile positive testing site can be compared with the other nearby sites and then statistically evaluated for spatial relationships between the positive testing area, the vegetation type and the related land use feature. In theory, a trap site that primarily consists of a particular vegetation type suspected to be associated with west nile vector ecology, should then chow a certain degree of high correlation statistically based on this analysis method.
The numeric values in the table represent degrees of correlation, i.e. with 1.00 representing 100% correlation, and 0.50 = 50% correlation, etc. This means that, once again in theory, those trap sites with the desired features (fairly low canopy-related light penetration as inferred by vegetation zone/area types for the trap site based on RS data and images) will be the mostly likely carriers of vectors associated with west nile, or carriers that are positive testing for west nile.
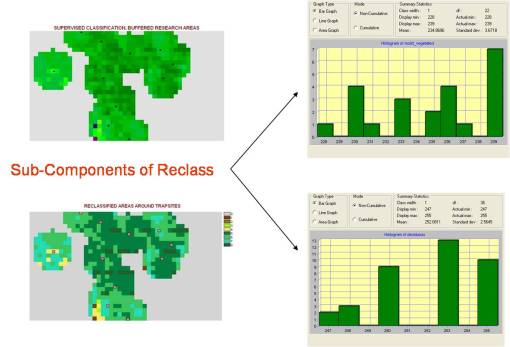
We could in theory interpret the counts of each of the reclassification subcomponents as forming a “signature” for other wetlands. This is uncommonly the case, however, due to variations and diversity of wetlands elsewhere in the local region. Still, parts of this histogram may serve as wetland indicators, such as the commonness of certain lower value cell value ranges or the absence of cells found in specific midrange values, or the ratio between the counts of one specific cell range and the counts of another cell range may be used as an indicator. To determine if such is the case would require repeated testing of this remote sensing model in other areas with similar phytoecologic findings (work yet to be completed on other Acer saccharum specified region with combined deciduous forest and westlands-moist-vegetation region ecosystems).
When we look at the distribution and counts of each of the reclass forms developed using the Reclass of a Reclass, in order to reduce the variability in possible vegetation zone definitions used for this analysis, we find the areal values for each of these zone types to be as follows. This dataset is of course much easier to apply to disease ecology analysis and prediction modeling than the more complex images above of NDVI such as the biodiversity or species richness imagery.
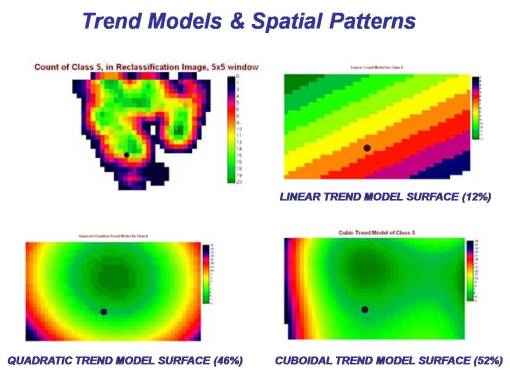
Reviewing trap locations in relation to the various zone patterns, it is apparent that Classes 5 and 6 represent the best numeric/spatial relationship to the point representing the positive testing site for this area. Class 5 -Deciduous (cover type) in fact demonstrated the best relationship spatially and numerically, followed very closely by Class 6 – Moist Vegetated. [See table of correlations for more.] We can then use this outcome to develop a prediction model for where the west nile vector is most likely to be found.
For prediction modeling, there are several ways we can go with the various equations used to produce prediction modeling equations. The simplest thing to do is to start with a single equation rather than engage in some method of combining such equations to determine if a more complex spatial relationship exists. (In 1997, I used this to model a landsurface with flowing water features, demonstrating that a linear 2-dimensional surface with a 1-dimensional equation best predicts water flow across a fairly slowly changing, flat, 3-dimensional surface, whereas the 3-D topographic features such as mountains, valleys, etc., are responsible for only 5% to 15% of the water flow across a surface.)
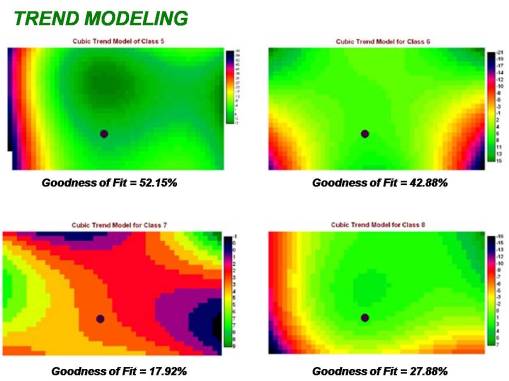
The following prediction modeling outcomes were produced. These methods for modeling spatial distributions were applied to the just the results of the Class 5 data, the area immediately adjacent to the house and yard. Linear, quadratic and cuboidal trend modelling are most often applied in this method for evaluating a land surface.
The site with its point (the positive test site) positioned closest to the center of the highest risk portion of these images does not necessarily represent the best correlation due to adjacent area patterns. Applying this to the rest of the zone patterns evaluated, in particular the higher risk areas (classes 6 through 8), we find that other areas produced the results below, many demonstrating very little correlation with the positive testing site.
The applications of this method of disease modeling and outbreaks prediction are fairly basic. At a macrogeographic scale, we can use this method to define where the most likely areas to be able to harbor the vectors and animal hosts for west nile, and then use that information to determine where traps should be set and where more expensive monitoring tools may be placed such as the placement of chicken coops, in which the chickens are used to test for vector bitee by species that are already harboring west nile. At the localized, much smaller geographic scale, we can use this form of analysis to determine our trapping activities and behavior when a high risk small area has been identified, of we can use it to monitor sites where known positive west nile cases have been found to exist.
Whatever the reason selected for using this method of studying the ecology of west nile and its host-vector relationships, the use of this method is valuable in not only monitoring west nile and detailing the results of the various surveillance activities we engage in, but also in west nile prediction modeling at a fairly local, small area level. The use of a remote sensing analytic method in combination with linear/transect modeling of disease ecology behaviors and grid analysis of specific high risk areal settings, GIS becomes a much more valuable tool than noted currently for much of the west nile analysis being performed using mostly non-correlative spatial point data.
The large and often costly efforts being put into west nile surveillance databasing and analysis using GIS at the medical epidemiological level does have better research avenues to take. This data requries a much more detailed and sophisticated analysis to be performed using ecological data in combination with physiographic and demographic/epidemiological data. Although more time is required to engage in such forms of analysis, a simple increase in work activities of 25% may be all that is needed to produce end results that are worth more than the added results produced by simply tripling your trap sites throughout a larger region for an applications that is simply point-based. Combining the standard point analysis routines with these area analysis techniques will result in better, more productive and educational methods of researching the ecology of zoonotic diseases such as west nile. Such methods will result in better more preventive, cost-effective uses for the various surveillance techniques we spatial epidemiologists like to employ. These methods make more valuable use of out time, and employ more of the skills applicable to spatial epidemiology and disease ecology that enable us to produce better GIS findings.
Final Comment
My impressions about the potential applications or Remote Sensing to west nile surveillance is that this is a very valuable research method that may have applications to west nile and other similar mosquito-borne disease diffusion processes, or it may simply serve as a stepping stone to experimenting and hopefully testing the applications of remote sensing to disease surveillance.
For the most part, my remote sensing work has dealt with Landsat images of various generations, and the use of Normalized Difference Vegetation Indices (NDVI) work involving various research regions and opportunities. I still work occasionally with SPOT imagery, and make ample use of the various spatial data forms out there to engage in other projects every now and then, like an intense study of NDVI and AVHRR imagery in the analysis of phytoecology in relation to landuse, hydrography and west nile disease patterns.
Theory is just theory, until it is put to good use.
.
Other pages:
.