(C) 2006, 2010 Brian Altonen
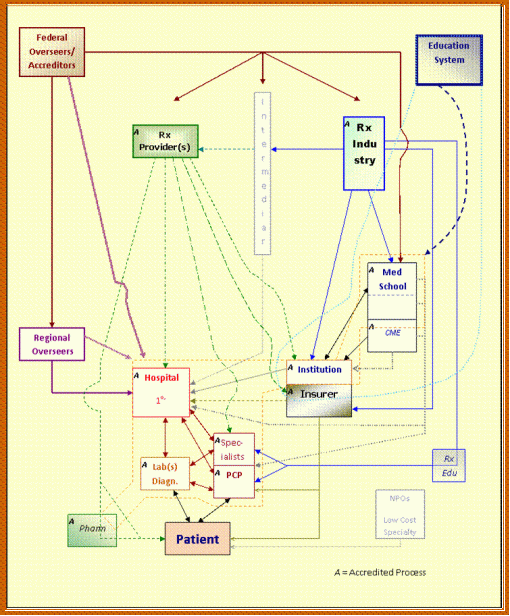
The philosophical ‘elephant’ in public health and medicine has its various components for researchers to contend with. The best place to study health is within the institutional or health care setting, for this is typically the best place to obtain the most accurate and detailed information pertaining to the health of an individual and the average health for the various groups of members that form the population. The health care system typically provides researchers with first hand recapitulations of visits, family history, labs, diagnostic testing results, any preventive activities to be engaged in, recommendations made by clinicians, and therapeutic plans designed by particular health care groups and programs. These different levels of health care have their own internal and external quality assurance practices that are engaged in and reported on, such that this usually takes place at some fairly consistent and ongoing level.
In the above flowchart, the one lone set of outsiders for this health care system consists of Rx Providers. This group consists of large scale industries that exist as groups or conglomerates of various businesses, each serving to provide its own one small part of the entire health care industry. The following depicts levels and agencies with measurements or QA activities in place:
Each of the different parts of this web of services makes regular contacts with various other service groups, QA programs, npos, Federal Agencies, and the like. On first glance we see the patient and the directly-linked health service provider for the patient forming the mainstay for this program. Together these two features serve as most crucial set of components found within this system. Without either, a system simply does not exist due to its singular nature. This process for evaluating the smallest component of the health care system is pretty much conisdered a true indicator of performance at both the clinical performance and non-clinical level. However, these events take place within the given clinical, hospital and office settings are often a consequence of activities that take place outside the patient-hospital-institution/insurer-medical school system delineated above (bordered by the yellow dotted line).
Most practices contained in the policies and procedures books for these health care providers are defined by the education system from which medical practice and therapeutics techniques have been defined. This education system (in the blue dotted line) overlaps with the health care providers system just enough to maintain some congruity between these two parts of the overall health care system. It is within the education system that agencies form and begin to develop their recommendations for treatment and the like, such as the American Association of Family Practitioners (AAFP) and American Academy of Pediatrics (AAP), which together define the childhood immunization recommendations, or the American Cancer Society and Amercian Academy of Obstetricians, each of which help to establish and update breast and cervical cancer screening recommendations on a periodical basis. Even when it comes to the recommended use of prescription drugs, there are some guidelines established and/or recommended by these special interest groups, included as well within the education system for this particular presentation.
The activities these professional agencies engage in have a direct impact upon all other activities that take place at lower levels depicted in the above flowchart. Nearly all hospitals or health care systems pay heed to the recommendations published by professional groups, although some may be delayed in engaging in such changes in practice. Most insurance agencies honor the professional recommendations of these agencies most of the time. Most of the allied health service providers, including independently operating pharmacists, pay attention to changes in protocols, especially when these changes become part of the health care agency they are affiliated with.
A major difference between the groups detailed so far in this flow of information related to practice is the level at which each of these groups is involved with the changes made in health care. All of these agencies at the clinical level are active participants, and therefore can easily measure any changes in performance that they make due to their education in the new measures, followed by the establishment of new policies by their institutions. They are for the most part, expected to engage to some extent in nearly all of these changes that are recommended by their health care institution. They are usually not expected to be told how to change their practice techniques by the other onlookers, such as the insurance agencies attached to their patient load and the largest and most passive group of overseers, pharmacy script providers. Instead, they are expected to receive recommendations on a regular basis, in particular by insurance agencies monitoring the health of their own particular patient populations.
A slight exception to the above involves Federal or Governmental overseers, in particular major insurers like Medicaid and Medicare. These two agencies do have the ability to monitor their patient health in general and take an active role in impacting each of the health care providers or institutions involved in these activities. This is typically for the betterment of population and patient health, but may also be employed due to underlying financial and manpower reasons. When such changes are made, they are expected to occur fairly rapidly, such as a change in protocols for treating a particular medical condition or the addition of a new monitoring measure developed for an unexpected rise in particular disease states.
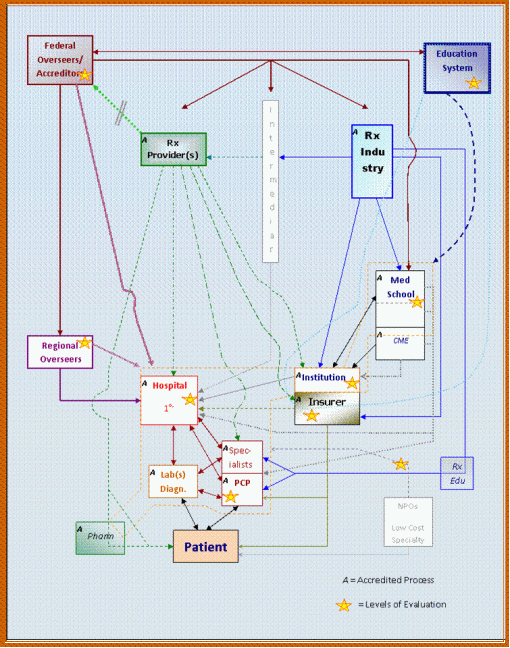
The following depicts these particular features pertaining to the flow of medical and population information in the current system.
For the most part, regional overseers and federal or government overseers remain pretty much active only as passive observors regarding the health care system and its provisions. Another fairly inactive onlooker in this schematic is the pharmaceutical industry in general, which even though it does often participate in particular studies, such as the development of a new cancer drug regimen, it still has certain corporate-based protective measures already in place, with the goal of preventing the release of too much information for one or more of its activities on a fairly regular basis. For this reason, RxProviders remain the only level at which extensive engagement of both corporate and non-corporate or public health related research can be fully engaged in. For much of the past decade, as these large companies developed their sizeable patient load and related datasets, most of this information has served as corporate purpose for use in measuring such things as income, rates of economic and institutional growth. These measures are rarely employed for uses related to a public health purpose.
One major feature of RxProvider data is that it exists at such a level and rank in the health care system that it can often be difficult for such a level in this hierarchy to have much of an impact far below its level in the preventive health scheme. It is possible that because this level of monitoring is simply passive in nature, and relies mostly upon retrospective data provided about patients and patient health in the form of claims-generated datasets, that little has been done by bearers of this information to develop this data into important information sources. To engage in such a process, it helps to understand that the best use of this information is going to population health related, with subgroups for studies used to define failure or success developed based upon insurer, location and general demographic datasets. Most likely, the main level at which this information impacts population health is at insurer and institutional levels, with some possibilities of pharmacal corporation and special interest group involvement as well.
This leaves us with the question–how can such measures best be developed and utilized to the best of their applicability, at the population health level?
The answer to this question is pretty much the reason the “Elephant” for this series of essays was observed, analyzed and then tackled as an important public health feature not yet placed into the limelight.
The Project Design
Due to the flow of information at the the RxProvider level, there is limited availability of case or medical records data. All work has to be producible using claims and script data. This section is an introduction to a methodology developed for this type of work from 2004 to 2006 pertaining to use of a National pharmacal database for PIP/QIA-related QA research.
The goal of any project developed for a measurement of the above mentioned RxProvider group level work has to be to produce a series of measures that can be directly linked to some reputable program or technique for measuring population health. For this reason, traditional population health measure methods such as HEDIS and PIP/QIA-related work were used to define the first part of this analytical process, followed by the development of anoth series of measures with reputations linked to well documented medical findings, such as the findings used to design and implement preventive health programs that follow a certain series of recommendations or guidelines. The final step in this development of a program for measuring population health outcomes involves the development of special measures determined to meet the needs of a special interest group or agency’s requirements, often assisting these groups in improving their overall internal health promotion process.
A preliminary review of data sets and past projects mimicking the types of work discussed resulted in the development of a methodology for monitoring population health based on a large population base and the use of primarily claims and script data by the development of a three tiered research process that engages in the following major stages:
- the research of population health based on HEDIS, and PIP/QIA measures already in place, with an emphasis on those studies that somehow involve the use of pharmaceutical products.
- the research of population health based on non-HEDIS measures, that may or may not be PIP- or QIA-related and which may or may not mimic HEDIS methodologies.
- the research of population health based on non-HEDIS measures that may mimic certain HEDIS-like features or methods, and which are engaged in due to special needs of the agency requesting this type of research.
- the research of population health based on methods that involve quantifiable outcomes related to one or more policy or guideline related procedures
The first stage pertains to the identification of 19 to 25 possible HEDIS topics that could be measured, based upon standard HEDIS protocols and methodologies. Each of these has a HEDIS-derived portion of research, followed by a comprehensive review of script and claims data to identify and measure the frequency of practice for a variety of human health behaviors, including activities regarding compliance, prevention and standard treatment protocols. One possible goal related to this stage of the work is the establishement of series of 50 major study sets (sets of questions with outcomes related to a single topic or purpose), each of which expected to be seen with each report, i.e. 18 HEDIS (i.e. HTN and HL script drug utilization rates, or frequency of antibiotic misuse for URI) + 25 HEDIS related (i.e. smoking cessation related prescription drug use, or chlamydia related need for medication) + 7 absolutely non-HEDIS (i.e. STD treatments in children 10 to 18 yo). An additional 15 to 25 additional study sets can then be added to agencies in need of lengthier reviews.
The second stage includes a series of measures that were directly related to other HEDIS measures already engaged in by the agency involved. These measures have other forms of documentation supporting their purpose and needs (guidelines, published treatment recommendations, etc.). The steps in developing this part of the study not only resulted in a series of measures that mimic other forms of HEDIS measurements already in place, but for different disease types or ICDs, but also resulted in the design of a series of fairly specific measures developed to measure performance and population health status for two or more dinstinctly different conditions. This methodology mimics a HEDIS measure that is already a standard implemented on an annual basis, but focuses on issues important at various other levels in the public or community health world, sometimes at some community level, other times at a sociological or cultural level.
The third stage provides information specific to the needs of the contracting agency requesting such work. These would be of studies specific to that population, for example, a study of the incidence of missed cancer screenings experienced by a particular socioeconomic group. In preparation for this level of research, a number of measures have to be developed which meet the requriements for population health indicators. These requirements focus on such primary features as age or age group, gender ethnicity, hispanicity, and socioeconomic status (SES). All of these studies require adequate documentation of the need for such work published in traditional medical references or journals.
The fourth stage is the development of measurements based on highly respected policies and guidelines. These policies and guidelines may relate to a sequence of activities required in order for a specific drug-related intervention activity to be developed, or may involve the use of some specific form of treatment or therapy that involves a multistep evaluation process with defineable precedents and required clinical-, diagnosis- or lab-related activities.
Approximately 30 measures can be developed for the first part of this methodology. These measures are directly related to 20 HEDIS measures. An additional 20 to 30 measures were identified as Stage 2 and Stage 3 types of measures. Since a number of these measures can actually be subdivided into multiple measurements, this increases the counts of measurable outcomes to around 60 to 75. (For example, the Asthma measures identified for this type of project pertain to three age groups according to HEDIS standards, with one or two more additional age groups that can be analyzed as well.) Additional insights into these methods are provided in part in the section detailing the 19 HEDIS measures reviewed and tabulated in terms of the findings for this project. The final series or group of measures is focused on special needs and “hot topics” in the field of medicine for the time. The potential for these measures is quite significant, although it is important to not over-empahsize this part of the program development process. For a typical study which included sopecial groups topics and measures, it is anticipated that at 30 to 40 such measures should be employed, in order to maintain a balance with the already well establishement teachings of measuring population health such as HEDIS (n=19-25) and PIP/QIA/guidelines (n=40-50).
It is also important to keep in mind that a well balanced series of measures needs to be developed for each activity and maintained throughout these processes. The HEDIS and non-HEDIS measures developed for this work consist of questions that serve as measures of just patient performance, others as measures of physician-patient performance, and perhaps a few just as indicators of PCP performance.
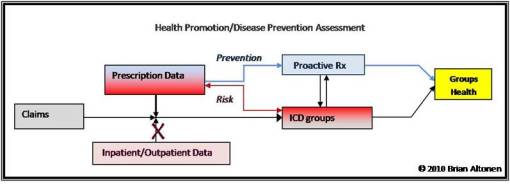
The following research process will be referred to throughout the remaining parts of this work. The goal of this work is to produce a project with a standard report format, using a methodology I developed based on HEDIS and non-HEDIS methods which I term “Groups Health” measures. This details a method by which the health of various groups or subgroups of a given population can be analyzed and the overall population health for a program defined based on outcomes. Since many of these methods employed for this work are repeatable, a certain amount of cross comparisons can be made with other programs undergoing the same sort of review. (Such reports lack corporate names and other identifiers for these other programs under review.) The meaning of this flowchart is defined elsewhere on other pages.